June 11, 2020

“We call this ‘Disrupting the System’ because the system is broken.”
– Dana Dornsife, CEO and Founder, Lazarex Cancer Foundation
“We acknowledge that this is a journey not a destination as the issues that are preventing patients from accessing treatment through clinical trials are immensely complex. But we know that in order to make a difference, we all must come together.”
– Lyndsay Harris, Director of Corporate Social Responsibility and Diversity, Medidata.
Summary
We face an urgent and critical moment in time. COVID-19 is once again revealing the health disparities that have existed for decades, just as social unrest over racism is prompting us to revisit how we operate as a country on all levels including healthcare. It is more apparent than ever that we must do better to engage, improve access and an environment of inclusivity.
We have known for years that minority communities are rarely if ever included in clinical trials and that the system is in desperate need of a disruption and overhaul. The disparities we see in this part of medicine are shameful and progress in this arena is far too slow. A mere 5% of cancer patients enroll in clinical trials, about half of all cancer trials fail due to lack of enrollment.
Fixing this is a matter of life and death for all of us, no matter the color of your skin or your ethnic background. The American Cancer Society estimates that this year, there will be more than 1.8 million new cases of cancer diagnosed and more than 606,000 people will die from cancer so we need to disrupt the system to find bring about much-needed change and save lives.
Those are the challenges that have inspired Lazarex Cancer Foundation to hold this event for three years running, bringing together some of the biggest disruptors and innovators in the fields of oncology, patient advocacy and public policy in the hopes of finding real, sustainable ways to change the current cancer research landscape to make it more accessible to ALL patients. We can’t let the barriers in the path become the path. At Lazarex Cancer Foundation, we are committed to thinking outside of the box and making an impact, so through these panel discussions, our goal is to provide a platform for provocative dialogue, which we hope will inspire positive change, prompt systemic improvements, more inclusivity and innovative solutions.
Because of the pandemic, this year’s event was virtual for the first time. Hosted from the National Press Club in Washington, D.C., we featured guests virtually from around the country. Despite the remote nature of the event, it served as a lively, educational and inspirational forum to spark disruption and promote new, innovative, cutting edge ideas to increase clinical trial participation, access, boost minority enrollment and retention, remove barriers and enhance equity and innovation.
We can’t fix the clinical trial system if we don’t disrupt it. Here’s a recap of this year’s event.
How COVID-19 is disrupting clinical trials
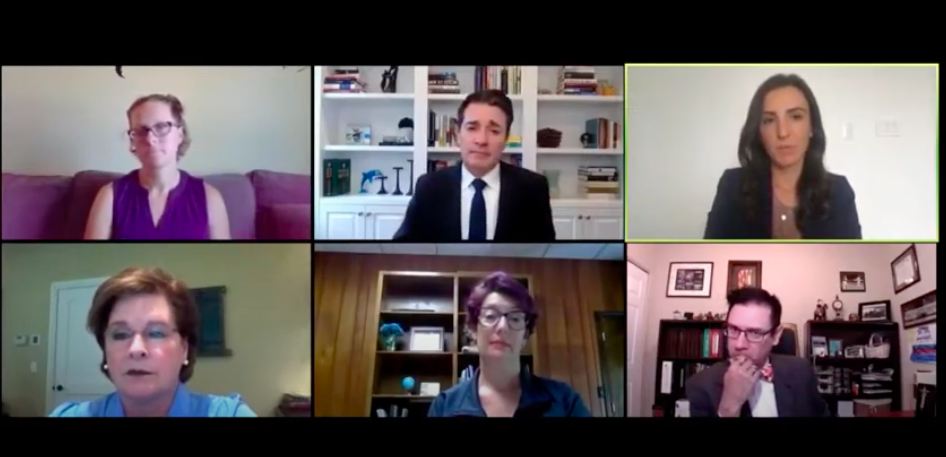
Moderator: John Whyte, MD, MPH, Chief Medical Officer, WebMD.
Panelists:
- Hala Borno, MD, PhD, a medical oncologist focusing on genitourinary malignancies at the UCSF Helen Diller Family Comprehensive Cancer Center.
- Jackie Kent, Executive Vice President, Head of Product at Medidata
- Becky Stephens – Currently being treated for Stage 4 breast cancer
View the video recording of this panel
Panelists opened by acknowledging that COVID-19 is affecting all aspects of patient care including clinical trials. Dr. Hala Borno, a medical oncologist at the UCSF Helen Diller Family Comprehensive Cancer Center explained that researchers are trying to do what they can to manage the complexities of navigating trials during a global pandemic by working to minimize participants’ activities to limit their exposure to the virus. That involves transitioning more processes to telehealth than ever before, which has both benefits and downsides.
Panelists agreed that increased regulatory flexibility because of the pandemic is also allowing for more use of technology to keep patients from dropping out of trials but they all worried that increased reliance on technology for telehealth and virtual health could end up widening an already sizeable opportunity gap, worsening outcomes or increasing adverse events among those with connectivity or internet literacy challenges.
“I do believe in rethinking the burdens we place on patients. I’m a big proponent of using technology to make patients lives better. But it is not necessarily accessible for everyone and as we design solutions and resources for patients we need to ensure they are accessible for the most vulnerable,” Dr. Borno explained.
Becky Stephens, who is currently part of a clinical trial for her Stage 4 Breast Cancer, says the pandemic finally allowed her to fulfill more of her trial responsibilities close to home, something the mother of two has long wanted. She says she hasn’t had any problem transitioning to more virtual contact with her trial doctors either but says she is tech savvy and has the necessary resources. “It’s not an uncomfortable situation for me but I could see that for someone who isn’t comfortable with technology that it could be stressful and hard,” Stephens said.
Panelists also discussed the lack of minority representation in clinical trials – numbers that haven’t moved much in two decades. “If we are talking about being disruptive and provocative, let’s look at the data,” Whyte says. “In cancer clinical trials, the number of people of color is extremely low and not increasing.”
Jackie Kent Executive Vice President, Head of Product at Medidata says she does see pharmaceutical companies working to change that. “It will take a little bit of time,” she says. “But companies are coming to us on a daily basis and asking us to help provide them information to not only design studies differently but to conduct feasibility and enrollment in different manner to find the right populations to encourage and enforce their companies to execute their trials with the correct diversity in their populations.”
Dr. Borno says ultimately, to truly change the system, you can’t just enroll patients coming in the door of your center. “When you are thinking about disrupting this area and accruing more diverse populations into clinical research, it can’t only be within a health system. It can’t only be an in-reach intervention for the patients you see in clinic. It really needs to be an outreach, more population-level and community based, getting the patients you don’t traditionally see at your cancer centers.”
Disrupting Engagement – The Community Voice in Action
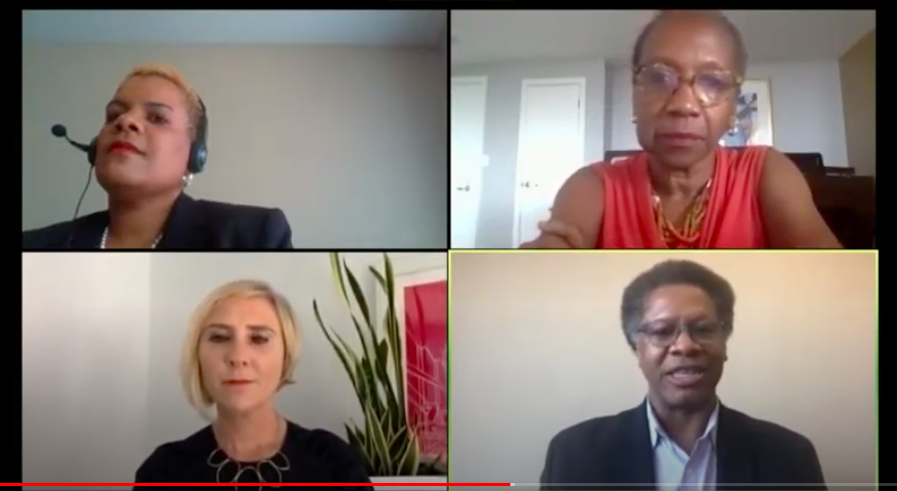
“You won’t get people to come if they don’t believe in the message and the messenger.”
-Loretta Sweet Jemmott, PhD, FAAN, RN, Vice-President of Health and Health Equity at Drexel University and Director of the Community Impact Program.
Moderator: Naima Gibson, Medidata Marketing Director
Panelists:
- Loretta Sweet Jemmott, PhD, FAAN, RN, Vice-President of Health and Health Equity at Drexel University and Director of the Community Impact Program.
- Robert Winn, MD, Director of Virginia Commonwealth University (VCU) Massey Cancer Center.
- Laura Holmes Haddad, Author, speaker and breast cancer survivor who attributes her survival to clinical trial participation.
View the video recording this panel
Robert Winn, MD, Director of Virginia Commonwealth University (VCU) Massey Cancer Center, opened the second panel with the observation that COVID-19 is spotlighting disparities in healthcare that some are only just now noticing – even though they have always existed, especially as they relate to clinical trials. He says to increase minority participation in clinical trials you must focus on prevention, engagement and screening.
“It has to be about more than drugs. It’s about people,” Dr. Winn explained. “While we are fascinated by the new drugs that we discover, it’s really about community engagement, prevention efforts, access to good foods…education even matters.”
Lazarex has created the iMPACT program, a 3-year nationwide program to increase cancer clinical trial enrollment, retention, minority participation and equitable access. The program aims to get cancer drugs to market faster and finish oncology clinical trials on time and on budget. Lazarex is now running Community iMPACT a pilot study with Drexel University School of Public Health in west Philadelphia with Loretta Sweet Jemmott, PhD, FAAN, RN, Vice-President of Health and Health Equity at Drexel University and Director of the Community Impact Program.
Dr. Jemmott says she is working with iMPACT to do that by listening to community voices that have long been voiceless to understand their concerns and then creating videos to counteract those worries. “This whole initiative is community-led and community-voiced,” Dr. Jemmott says. “It is time for us to do something different, disrupt the system and make the change. What we have learned in this process is this takes time. But hearing the voices of the community is crucial.”
Laura Holmes Haddad, a cancer survivor, says this work is incredibly important because it literally saves lives, including hers. When standard treatment failed against her stage 4 inflammatory breast cancer, she was given 2 years to live at the age of 37. The mother of 2 credits a clinical trial with saving her life. “I would not be standing here today if it weren’t for a clinical trial,” she explained. But Holmes Haddad says this route has not been easy. She says it took her entire community to make this possible for her including friends and strangers who donated hotel and mileage points and made trips with her to her trial site when she was too sick or tired to do it alone. “As much as it was a lifeline, it was so difficult emotionally and physically,” Holmes Haddad says, adding that it was still very much worth it because after 6 months the trial regimen began to work and 5 years later she remains cancer free. “Now I try to speak out and try to really encourage others to seek out clinical trials and to ask about clinical trials because it saved my life.
Disrupting the Payment Platform – Exposing the Barriers to Patient Access
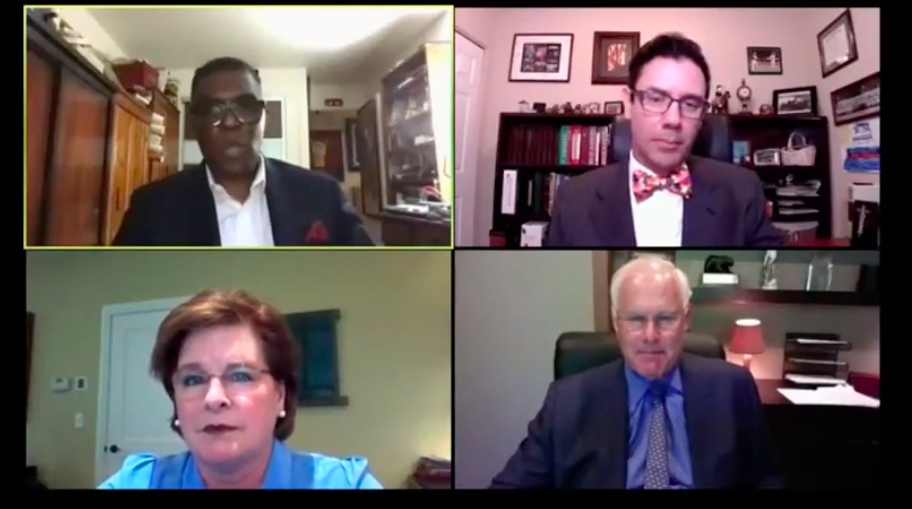
“What kind of healthcare system do we have? I think it’s a fundamentally broken one to be very honest.”
– Mark R. Laret, President and CEO of UCSF Health.
Moderator: Dana Dornsife, CEO and Founder, Lazarex Cancer Foundation
Panelists:
- Glenn Ellis, MPH, Medical ethicist and President of Strategies for Well-Being.
- Mark R. Laret, President and CEO of UCSF Health
- Fabian Sandoval, MD, CEO and Research Director of the Emerson Clinical Research Institute.
View the video recording of this panel
Dana Dornsife, Founder of the Lazarex Cancer Foundation moderated the Third Panel focused on creating a platform of equitable access to trials, removing barriers and looking at how the payment system – also known as the payer mix, creates disparities based on who can access the system and where. “The system is broken. It doesn’t work for everyone. It only works for some,” Dornsife explained.
Panelists addressed difficult and longstanding barriers and disparities that Ellis said are evident in a wide range of statistics including a shocking one he found upon researching data from the Association of American Medical Colleges.
“We have to do something about the diversity in the field of research and in the medical profession,” Ellis said. “Today as we speak there are only 500 black medical students in all the medical schools in this country. Five hundred. That’s it.“
“Glenn, that gives me tremendous pause,” Dornsife said.
Ellis agreed, adding, “Imagine how helpless – imagine what it feels like to be in a country during the middle of a pandemic with this coronavirus and you can’t even get to a primary care doctor. So this is not about us trying to figure out how to get to California from Philadelphia to get in a clinical trial. People can’t even get out of their neighborhoods. Where are they going to go?”
Panelists debated how to incentivize the healthcare system to embrace more diversity and stressed that the problem is compounded by the fact that people’s insurance determines how they are treated. Everyone agreed that Medicaid patients, for example, don’t generally have access to the latest and greatest in clinical trials if they aren’t offered at the location where they’re seen, and their insurance is accepted. Dr. Sandoval says solutions can be found in allowing more providers to be picked to be part of clinical trials and grooming more young providers to become researchers. “We need to not just have research performed at large academic settings but community clinics as well,” Dr. Sandoval suggested.
Mark R. Laret, President and CEO of UCSF Health agreed, adding that he doesn’t believe you can’t fix the system unless you take on the whole model of an employer-based healthcare system. “This patchwork quilt that works great for some but is a complete nightmare for so many others, particularly poor people and people of color, is a tragedy and a travesty in my mind,” he said.
Panelists also agreed that it’s long past time to address systemic and structural racism and white privilege in the healthcare system. “We have to do something about diversity in the field of research and in the medical profession,” said Glenn Ellis, MPH, Medical ethicist and President of Strategies for Well-Being. “The question is going to be – Who do we want to be as a society? Who do we want to be as a medical field?”
Laret agreed. “We in the health care system have to take on this whole issue of our long history of marginalizing African Americans in particular but it cuts across all racial and ethnic groups,” he said. “We start there with that and leverage this moment in time to do something different. Economics is one element but there is a fundamental structural racism piece.“
Celebrating the Disruptors: the 2020 Lazarex Disruptor Awards

Fabian Sandoval, MD, CEO and Research Director of the Emerson Clinical Research Institute.
Through his Institute, Dr. Sandoval works to raise awareness about clinical trials and is deeply dedicated to serving patients who need these trials to survive. Through the creation of Emerson Clinical Research Institute and his show on Telemundo: “Tu Salud, Tu Familia” he has been a disruptor in thought and action who works tirelessly to break barriers to close the gap, give a voice to many in the Latino community and build awareness of clinical trials to create more equitable access.
“We have to do boots on the ground everyday activity and the work we do is done with love and care and education because not every patient that needs the medication or needs these treatment knows this is an option,” Dr. Sandoval says.
John Whyte, MD, MPH, Chief Medical Officer at WebMD
Dr. John Whyte has answered the call to disrupt the system in both thought and action. When Dr. Whyte served as the Director of Professional Affairs and Stakeholder Engagement at the Center for Drug Evaluation and Research at the U.S. Food and Drug Administration, he helped change the system by changing FDA guidance to allow patients to be reimbursed for travel costs associated with clinical trials. He took a leadership role in shepherding that monumental change at the federal agency and he continues to show partnership and solidarity in lending his voice to help expand the conversation to increase enrollment in cancer clinical trials, minority access and retention.
“What I try to do working with you and others is ask – how do we change the system,” Dr. Whyte explained. “The whole goal of changing the system is to do it in a way that benefits patients.”
Recent Successes
Lazarex Cancer Foundation has been engaged in intensive efforts at the state and federal levels to alleviate financial burdens faced by patients who wish to participate in clinical trials.
June 2007 – December 2019, Lazarex assisted over 5,000 patients with patient navigation and financial assistance for FDA clinical trial participation.
March 2020 – Gov. Tony Evers signs bill SB 489 into law in Wisconsin to address economic barriers faced by cancer patients in accessing clinical trials. Bill passed on voice voices in both the Assembly and the Senate with bipartisan support. Read more here.
December 2019 – Illinois lawmakers pass new law – SB1711 – to help cancer patients access potentially life saving clinical trials. Read more here.
September 2019 – New Texas law helps cover some costs for cancer treatment patients. Read more here.
Action Steps
The panels discussed several key elements that are necessary to improve access and participation in clinical trials going forward including:
- We need to continue to raise awareness of the lack of participation and diversity in clinical trials.
- Partnering with communities to improve awareness of and access to clinical trials makes a difference and we need to do more of this work.
- Legislative action is needed in more states to remove economic barriers and address reimbursement and improved access for clinical trial participants.
- The pharmaceutical industry must participate in the reimbursement process.
- The payment platform must be disrupted – to expose barriers to access and begin to remove them.
If you missed this event – or would like to watch any of the panels again, you can find them on YouTube and Facebook
Find out more about Lazarex at Lazarex.org and Medidata at Medidata.com
Lazarex is the only 501(c)(3) public charity that focuses on helping patients with all types of cancer, of all ages, from all walks of life. We remove the barriers to clinical trial participation for advanced stage patients seeking life through FDA clinical trials. In doing so, we are proving to federal and state lawmakers, leaders of the biopharmaceutical industry, heads of cancer clinics and the top academic institutions that the simple act of reimbursement increases enrollment rates and minority participation and creates a platform for equal access that benefits us all. When you reimburse a cancer patient for the travel cost to get to a clinical trial, patients will participate, stage engaged in their fight and more will have a chance to live.
During this COVID-19 crisis, Lazarex continues to assist cancer patients with FDA trial navigation, reimburse trial-related travel expenses, and work with industry stakeholders to ensure ALL cancer patients have access to clinical trial treatments. You can help support our work and the patients we serve HERE.
Click here to see the impact the global pandemic has had on cancer clinical trials – from Denver’s ABC affiliate
Medidata Solutions is an American technology company that develops and markets software as a service for clinical trials. These include protocol development, clinical site collaboration and management; ran randomization and trial supply management; capturing patient data through web forms, mobile health devices, laboratory reports, and imaging systems; quality monitor management; safety event capture; and monitoring and business analytics. Headquartered in New York City, Medidata has locations in China, Japan, Singapore, South Korea, the United Kingdom, and the United States. To learn more, visit www.medidata.com




